CBD and the Endogenous Cannabinoid System – Part 2
![shutterstock_1137745376 [Converted]](https://innocanpharma.com/wp-content/uploads/2020/05/shutterstock_1137745376-Converted.jpg)
The Endocannabinoid System – A closer look
Since the confirmation of the existence of the Endocannabinoid System (ECS), much work has been done to try to unravel this powerful yet convoluted feedback system. When one considers the enormous complexity of the Central Nervous System and the functions of our organs and peripheral nervous system, it becomes evident that the endocannabinoid system could affect behavior and our bodily functions in an almost limitless number of ways. Simple generalizations of what will happen when CB1 and CB2 receptors are globally turned on or off are not feasible. As such, it is clear that researchers have only scratched the surface of the endocannabinoid system and there is so much more to learn.
In simple terms, what is understood at this time is that the ECS works to maintain homeostasis within the body. The ECS is comprised of cannabinoid receptors, endogenous cannabinoids (endocannabinoids), and the enzymes responsible for the synthesis and degradation of the endocannabinoids. When homeostasis is disrupted in any part of the body the appropriate endocannabinoid will be synthesized and released to bind with the appropriate cannabinoid receptor. After it binds it is quickly degraded by one of the enzymes listed earlier. This is, as stated, a simple description of what happens when the ECS is activated. What actually happens is still the subject of considerable research, but a great deal has been learned in the last 20 years. This article will focus on some of the key findings during this time, starting with the endocannabinoids themselves.
Endocannabinoids
Although endocannabinoids were the last to be discovered, it was the research being done with phytocannabinoids, specifically THC that led to the eventual discovery of the endocannabinoid system. Both 2-arachidonoyl glycerol (2-AG) and arachidonoyl ethanolamide (anandamide) or (AEA) are the best-studied endogenous cannabinoid ligands. Endocannabinoid ligands are unsaturated fatty-acid ethanolamides, glycerols or glycerol ethers.
The presence of other endocannabinoids is fairly certain, however the biology of these compounds are not as well developed as that of AEA and 2-AG, so they will not be discussed in this article. For example, N-homo-y-linolenoylethanolamine and N-docosatetr-aenoylethanolamine have been shown to bind to the cannabinoid CB1 receptor with high affinity. Other endocannabinoids identified to date include N-arachidonoyl dopamine (NADA) and virodhamine. Both have been shown to bind to CB1 receptors. (1)
The pharmacological characterization of AEA and 2-AG, show each possessing distinct properties. AEA is a high-affinity, partial agonist of CB1 receptors, but is almost inactive at CB2 receptors; whereas 2-AG will act as a full agonist at both receptors with moderate-to-low affinity. It has also been discovered that AEA and 2-AG are not exclusive to CB1 and CB2 receptors. It is now understood that they interact with other receptors as well, much like the phytocannabinoids cannabidiol. For example, AEA has been shown to activate the transient receptor potential cation channel subfamily V member 1 (TRPV1). TRPV1 is documented for its significant role in synaptic transmission and pain regulation. (2)
An important feature of these endocannabinoids is that their precursors are present in lipid membranes. They are synthesized upon demand by activation of certain G protein-coupled receptors or by depolarization. Endocannabinoids are liberated in one or two rapid enzymatic steps and released into the extracellular space. This contrasts with classical neurotransmitters that are synthesized ahead of time and stored in synaptic vesicles. (3)
Synthesis of Endocannabinoids
Despite similarities in chemical structure, 2-AG and AEA are synthesized and degraded by distinct enzymatic pathways. AEA is catalyzed from N-acyl-phosphatidylethanolamine (NAPE), by NAPE-specific phospholipase D (NAPE-PLD) or via other routes not involving NAPE-PLD. On the other hand, 2-AG is produced from diacylglycerol (DAG) by either DAG lipase (DAGL) α or β, although most if not all 2-AG mediating synaptic transmission in the adult brain is generated by DAGLα. The rate-limiting and Ca2+-sensitive step in AEA and 2-AG production, however, is the formation of NAPE and DAG, which are converted from phosphatidylethanolamine by N-acyltransferase and phosphoinositides by phospholipase C, respectively. (2)
Once released into the mostly aqueous intracellular space, endocannabinoids, which are lipids, are unable to diffuse freely like other neurotransmitters. How the endocannabinoids span the space is still not known, however a popular theory is they are transported by certain carrier proteins like fatty acid binding proteins and heat shock protein 70.
Figure 1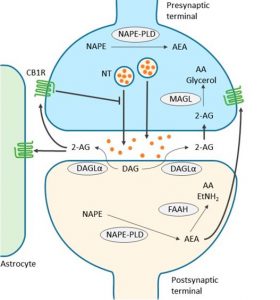 |
Endocannabinoids are produced from postsynaptic terminals upon neuronal activation. As the two major endocannabinoids shown in the scheme, 2-AG is biosynthesized from diacylglycerol (DAG) by diacylglycerol lipase-α (DAGLα), and AEA is synthesized from N-acyl-phosphatidylethanolamine (NAPE) by NAPE-specific phospholipase D (NAPE-PLD). As lipids, endocannabinoids, mainly 2-AG, readily cross the membrane and travel in a retrograde fashion to activate CB1Rs located in the presynaptic terminals. Activated CB1Rs will then inhibit neurotransmitter (NT) release through the suppression of calcium influx. 2-AG is also able to activate CB1Rs located in astrocytes, leading to the release of glutamate. Extra 2-AG in the synaptic cleft is taken up into the presynaptic terminals, via a yet unclear mechanism, and degraded to arachidonic acid (AA) and glycerol by monoacylglycerol lipase (MAGL). On the other hand, AEA, synthesized in postsynaptic terminal, activates intracellular CB1R and other non-CBR targets, such as the transient receptor potential cation channel subfamily V member 1 (TRPV1). Although endocannabinoid retrograde signaling is mainly mediated by 2-AG, AEA can activate presynaptic CB1Rs as well. Fatty acid amide hydrolase (FAAH) is primarily found in postsynaptic terminals and is responsible for degrading AEA to AA and ethanolamine (EtNH2). Although NAPE-PLD is expressed in presynaptic terminals in several brain regions, it is not clear yet whether AEA is responsible for anterograde signaling in the endocannabinoid system. Note that alternative routes exist for the metabolism of endocannabinoids, depending on the brain region and physiological conditions. Thin arrows indicate enzymatic process; thick arrows indicate translocation; blunted arrow indicates inhibition. (2)
A unique characteristic of endocannabinoids is that they travel backward (retrograde) across the synapse in the opposite direction that other neurotransmitters and bind with cannabinoid receptors located on adjacent cell membranes. Typically, when a neuron that releases a chemical neurotransmitter (say, GABA or glutamate) it is designated as “pre-synaptic”; the target neuron that expresses receptors for that neurotransmitter is “postsynaptic.” However, endocannabinoids will be synthesized in the postsynaptic cell and travel across the synapse to bind with CB1 or CB2 receptors on the pre-synaptic cell thereby inhibiting the release of various excitatory and inhibitory neurotransmitters, thereby regulating the transfer of their own incoming synaptic signals. (4)
Once endocannabinoids are taken up by the cells, they can be degraded through hydrolysis and/or oxidation. AEA is degraded by fatty acid amide hydrolase (FAAH) into free arachidonic acid and ethanolamine, whereas 2-AG is mostly hydrolyzed by monoacylglycerol lipase (MAGL) into arachidonic acid and glycerol; several other enzymes could be involved as well. Oxidation of both AEA and 2-AG could involve cyclooxygenase-2 and several lipoxygenase. (5)
Cannabinoid Receptors
Cannabinoid receptors (CBRs), as the name suggests are the part of the ECS that binds with cannabinoids. They are located throughout the body on cell membranes mediating endocannabinoid activity, which is believed to affect a variety of physiological processes including appetite, pain-sensation, mood, and memory.
CBRs are of a class of cell membrane receptors in the G protein-coupled receptor (GPCR) superfamily. This family of receptors may also be referred to as: seven-(pass)-transmembrane domain receptors; 7TM receptors; heptahelical receptors; serpentine receptor and G protein-linked receptors (GPLR). Like all GPCRs, cannabinoid receptors are helical in shape and contain seven transmembrane spanning domains.(6) There are currently two known subtypes of cannabinoid receptors, termed CB1 and CB2. However it is suspected that other cannabinoid receptors exist. These receptors will only be discussed briefly in this article as very little is known about them at this time.
Type and Distribution of Cannabinoid Receptors
CB1 Receptors
The CB1 receptor is expressed mainly in the central nervous system and peripheral nervous system. They are thought to be one of the most widely expressed Gαi protein-coupled receptors in the brain where it mediates neurobehavioural responses, but research has also shown they can be found in the lungs, liver and kidneys, as well as the pancreas and skeletal muscle. One mechanism through which they function is endocannabinoid-mediated depolarization-induced suppression of inhibition, a very common form of retrograde signaling, in which the depolarization of a single neuron induces a reduction in GABA-mediated neurotransmission. Endocannabinoids released from the depolarized post-synaptic neuron bind to CB1 receptors in the pre-synaptic neuron and cause a reduction in GABA release by inhibiting presynaptic calcium ions entry.
CB2 Receptors
The CB2 receptor is expressed mainly in the T cells of the immune system, on macrophages and B cells, and in hematopoietic cells. They also have a function in keratinocytes and are also expressed on peripheral nerve terminals. These receptors play a role in antinociception, or the relief of pain. In the brain they are mainly expressed by microglial cells, where their role at this time remains unclear.
Immune and immune-derived cells (e.g. leukocytes, various populations of T and B lymphocytes, monocytes/macrophages, dendritic cells, mast cells, microglia in the brain, Kupffer cells in the liver, astrocytes, etc.) are considered to be the most likely CB2 receptor targets of endocannabinoids, however, the number of other potential cellular targets is expanding, now including endothelial and smooth muscle cells, fibroblasts of various origins, cardiomyocytes, and certain neuronal elements of the peripheral or central nervous systems. (7)
Other Cannabinoid Receptors
The existence of additional cannabinoid receptors has long been suspected, due to the actions of compounds such as abnormal cannabidiol a synthetic isomer of the phytocannabinoid cannabidiol (CBD) that produces cannabinoid-like effects on blood pressure and inflammation, yet does not activate either CB1 or CB2. Recent research strongly supports the hypothesis that the N-arachidonoyl glycine (NAGly) receptor GPR18 is the molecular identity of the abnormal cannabidiol receptor and additionally suggests that NAGly, the endogenous lipid metabolite of AEA, initiates directed microglial migration in the CNS through activation of GPR18. (8)
Other molecular biology studies have suggested that the orphan receptor GPR55 should in fact be characterised as a cannabinoid receptor, on the basis of sequence homology at the binding site. Subsequent studies showed that GPR55 does indeed respond to cannabinoid ligands. (9) This profile as a distinct non-CB1/CB2 receptor that responds to a variety of both endogenous and exogenous cannabinoid ligands, has led some groups to suggest GPR55 should be categorized as the CB3 receptor, and this re-classification may follow in time. (9)
However this is complicated by the fact that another possible cannabinoid receptor has been discovered in the hippocampus, although its gene has not yet been cloned, suggesting that there may be at least two more cannabinoid receptors to be discovered. In addition to the two that are already known GPR119 has been suggested as a fifth possible cannabinoid receptor. (10)
Conclusion
As more research is completed it is clear that the Endocannabinoid System has many more working parts than originally believed. The discovery of novel endocannabinoids and cannabinoid receptors has raised questions that open the doors for new theories to be researched. This is an exciting time for those involved as it is thought the ECS holds the potential for new treatments of disease that previously had no new options.
In Part 3 of this series we will take an in depth look at what has been discovered about Cannabidiol, the non-psychoactive phytocannabinoid derived from Cannabis sativa as well as some of the latest discoveries connected to this super molecule. As Raphael Mechoulam said in a recent interview with Discover Magazine: “From my point of view, our body tries to fight every single disease in most cases with the immune system. It’s a very important system, but the immune system as we know it today doesn’t address some of the diseases. So, the body has other ways to try to fight these diseases, and I believe that the endocannabinoid system is one of the major systems through which the body tries to fight disease. So, maybe in a decade or so, we should be speaking of not only the immune system but the endocannabinoid [system]. It may be an additional way the body attacks disease states.” (11)
Articles Referenced
(1) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4120766/
(2) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5877694/
(3) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4789136/#R2
(4) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3997295/
(5) https://journals.physiology.org/doi/full/10.1152/physrev.00019.2008
(6) https://en.wikipedia.org/wiki/G_protein-coupled_receptor
(7) https://en.wikipedia.org/wiki/Cannabinoid_receptor
(8) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2865488/
(9) https://www.sciencedirect.com/science/article/pii/S1550413106000647
(10) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2190013/
More articles: